|
The American Academy of Pediatric Dentistry (AAPD) supports the preservation of primary teeth until natural exfoliation to maintain appropriate oral function and facial growth. Therefore, the maintenance of sound tooth structure and its supporting tissues remains a leading objective of pulp therapy in teeth that have been affected by caries or other injuries.
Currently, pulpotomy followed by a full-coverage stainless steel crown is the most frequently used treatment for carious lesions approximating a vital pulp in primary teeth. However, existing research has shown that an alternative technique, indirect pulp treatment, is a suitable option and results in similar or greater success rates than pulpotomy. A study conducted by Rosenberg and colleagues at New York University College of Dentistry demonstrates the efficacy of indirect pulp treatment in primary molars when using 2% chlorhexidine gluconate disinfecting solution and resin-modified glass ionomer liner. Additionally, the use of pediatric zirconia crowns provides an obvious esthetic advantage over stainless steel crowns. Indirect pulp treatment is indicated for treating large carious lesions that approximate the pulp and do not demonstrate signs or symptoms of irreversible pulpitis. Indirect pulp treatment involves removing the first layer of the infected dentin of a carious lesion, while leaving behind a thin layer of affected dentin to prevent pulp exposure. Next, a biocompatible liner is applied over the remaining affected dentin to stimulate healing at the dentin-pulp interface. Finally, the tooth is restored to form an adequate seal and prevent microleakage. Zirconia is a biocompatible material used widely for crown and bridge, implant, and endodontic procedures in adult dental patients. However, the use of zirconia for pediatric crowns was not available until recently. Zirconia crowns provide superior esthetics over preveneered and stainless steel crown options and they do not stain or chip. One important disadvantage to consider when using zirconia crowns, however, is that saliva and blood contamination can weaken the bond strength between the tooth and the crown. Some manufacturers have overcome this problem by developing try-in crowns to verify proper fit before the final crown is cemented. Furthermore, zirconia crowns may not be suitable in cases with significant crowding or space loss, as their size, shape, and fit cannot be altered. The following case presents a 6-year-old healthy patient with a large carious lesion on the lower left first primary molar. Indirect pulp treatment was completed after caries removal, and the tooth was restored with a zirconia crown. 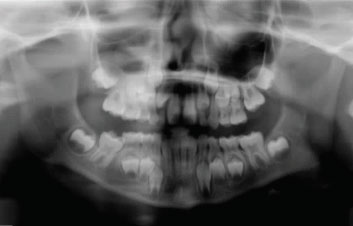
Figure 1—Panoramic radiograph 
Figure 2—Bitewing radiograph taken using a DEXIS digital x-ray sensor, demonstrating a large carious lesion on a mandibular left first primary molar with no evidence of inter-radicular pathology 
Figure 3—Anesthesia achieved with Septodont’s articaine hydrochloride 4% and 1:200,000 epinephrine, using a single-tooth anesthesia system (The Wand STA, Milestone Scientifi c) 
Figure 4—First primary molar following application of Sable Seek (Ultradent) caries indicator; infected dentin removed 
Figure 5—Application of Consepsis 2% chlorhexidine gluconate solution (Ultradent) 
Figure 6—Indirect pulp treatment: application of Vitrebond light-cured glass ionomer liner (3M) 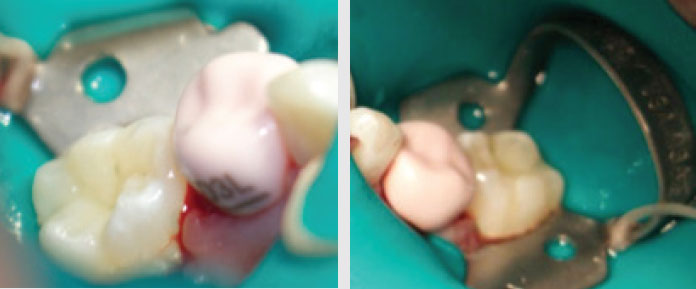
Figure 7-8—Buccal and lingual view of NuSmile ZR zirconia crown pink try-in size D3 
Figure 9—Immediate postoperative clinical photograph of NuSmile ZR zirconia crown cemented with NuSmile BioCem in mandibular arch, following verifi cation of occlusion. Due to space loss, the crown was cemented with a slight rotation; however, proper occlusion was still achieved as evidenced by articulating paper markings. |

Consepsis, a 2.0% chlorhexidine gluconate solution, is used for disinfection during endodontic procedures, as a fi nal rinse before canal obturation, and before pulp capping. Free of emollients that interfere with bond strength, it is formulated to minimize postoperative pulpitis and sensitivity by thoroughly cleaning and disinfecting the preparation prior to sealing and restoring. Consepsis also provides long-term antimicrobial activity, reducing the risk of recurrent caries.
ULTRADENT PRODUCTS
800.921.4806 ext. 169
https://www.dps.li/a/684–151

Sable Seek is a glycol-based caries indicator that identifies demineralized dentin in difficult-to-see places, such as under the overhanging enamel of Class I, II, or III preparations. Green Sable Seek helps prevent overexcavating deep caries, which can lead to pulp exposure, while Red Seek can be easily seen on dark dentin, providing a fast, effective way to locate calcified root canal orifices. With patient-friendly dyes, it is delivered precisely in a mess-free syringe.
ULTRADENT PRODUCTS
800.921.4806 ext. 169
https://www.dps.li/a/684–152






